Clipart tagged: ‘sulfur’
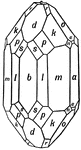
Acanthite
This figure, representing Acanthite, an orthrohombic crystal, shows a combination of the following forms:…
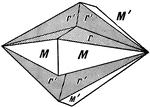
Arsenopyrite
Iron Sulpharsenide (Arsenopyrite), bounded by ∞P, {110} (M), and 1/3P∞̆, {013} (r), forms penetration…
Calcium Hyposulphite
This figure shows the combination of forms on calcium hyposulphite (CaS_2_O_3_ + 6 aq). If we assume…
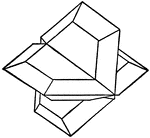
Chalcocite
The Copper Sulphide, Chalcocite, forms three kinds of twins symmetrical to ∞P, {110}, to 4/3P∞̆,…
![This figure represents a crystal of iron pyrite showing representatives of all the forms of the isometric system except the rhombic dodecahedron: ∞O∞ {100} (P); O, {111} (d); [∞O2/2], π{102} (e); [4O2/2], π{214} (s); 2O2, {211} (o); and 3O, {133} (t). It is shortened in the direction of one of the principal axes, which, together with the fact that only two of the three faces belonging to the forms s, o, and t are developed in each octant, gives to it a decidedly orthorhombic habit.](https://etc.usf.edu/clipart/86800/86858/86858_crystal-of-iron-pyrite-showing-representatives_mth.gif)
Crystal of Iron Pyrite Showing Representatives of All the Forms of the Isometric System
This figure represents a crystal of iron pyrite showing representatives of all the forms of the isometric…
Cube of Iron Pyrite
Cubes of Iron Pyrites frequently show a striation of their planes in one direction, which is perpendicular…
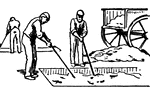
First Stage in Glue Manufacturing
This illustration shows the first stage in the manufacturing of glue. The material used for making the…
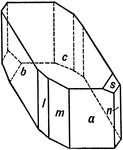
Hydrous Copper Sulphate
This figure reproduces a crystal of Hydrous Copper Sulphate whose forms are (c), (a), (b), (n), (m),…
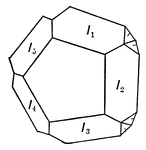
Marcasite
Orthorhombic Iron Disulphide (Marcasite) sometimes shows cyclic groups of five individuals, bounded…
![It has been observed that the pentagonal dodecahedron, [∞o2/2], π {201}, on pyrite may have but six of its twelve planes developed in such a manner a to produce an apparent rhombohedron.](https://etc.usf.edu/clipart/86800/86856/86856_pyrite_mth.gif)
Pyrite
It has been observed that the pentagonal dodecahedron, [∞o2/2], π {201}, on pyrite may have but six…
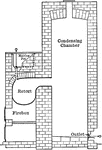
Sulfur Purification
"The sulphur is then further purified by heating it in iron vessels (retorts) without access of air,…
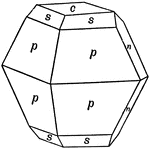
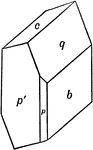
Sulphur
This figure shows an orthorhombic crystal of sulphur, showing the forms: (p), (s), (c), and (n).
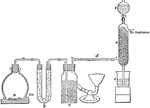
Sulphur Trioxide Preparation
Laboratory preparation of sulphur trioxide, an important step in the process of making sulphuric acid.…

Sulphuric Acid Contract Process
Diagrammatic representation of the sulphuric acid contact process. "Sulphur, or ores containing sulphur,…